Abstract
Background: D-dimer values in patients with SARS-2 COVID-19 infection are known to be elevated and have been shown to be a predictor for disease severity, mortality, and venous thromboembolism (VTE). Patients presenting with COVID-19 infection may endorse many of the same symptoms as patients with pulmonary emboli (PE) and the associated elevation of the d-dimer as a component of the inflammatory state has led to unanswered questions regarding the diagnostic utility of d-dimer testing.
Methods: Computed tomography (CT) scans of the chest with intravenous contrast were searched from a unified data platform between 1/1/2020 and 5/31/2022 across the Mayo Clinic enterprise. Imaging was matched to patients with COVID-19 if it occurred between 10 days before and 30 days after the laboratory confirmed COVID-19 infection. Next, patients were matched to laboratory records of d-dimer testing within 1 day of the imaging. D-dimer testing was performed using the HemosIL D-dimer HS 500 kit on the Instrumentation Laboratory ACL TOP instrument. A negative result is ≤500ng/ml fibrinogen equivalent units (FEU) with a lower limit of detection of 220ng/ml. The primary outcome was acute PE on the imaging study as determined by a validated natural language processing algorithm.
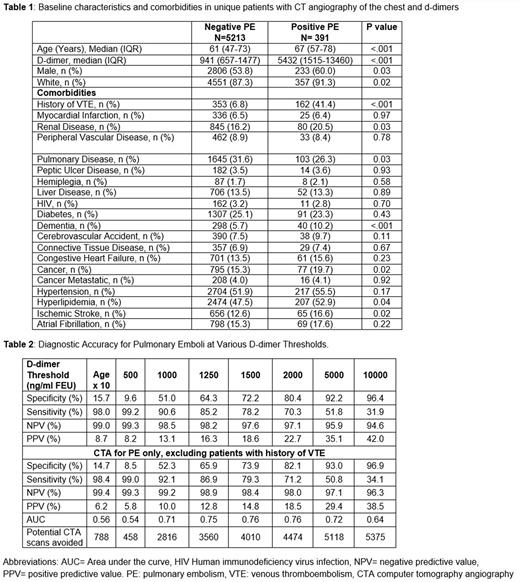
Results: A total of 6,747 d-dimer/CT pairs were examined in 5,971 unique patients with COVID-19. Pulmonary emboli were identified on 7.7% of CTA of the pulmonary arteries (n=6,222), 6.6% of CTA-other (n=241), and 3.9% of CTs with IV contrast (n=284). Patients diagnosed with PE were older (median age 67 vs 61, p<0.001), more likely to be male (60.0% vs 53.8%, p=0.03), white (91.3% vs 87.3%, p=0.02), and have dementia, cancer, hyperlipidemia, or ischemic stroke (Table 1). Median d-dimer values in patients found to have PE was 5432ng/ml compared 941ng/ml in those without PE. A prior history of VTE (by ICD-10 codes) was seen more frequently in the group diagnosed with PE (41.4% vs 6.8%, p =0.03). In the overall group of patients with CT imaging of the chest with some form of intravenous contrast, the sensitivity at the standard d-dimer threshold was 99.2% [negative predictive value (NPV) 99.3%], decreased to 98.0% with the age-adjusted cutoff (NPV 99.0%), and decreased further with an increased d-dimer threshold of 1000ng/ml (NPV 98.5%) (Table 2). Due to the strong association seen between patients with a history of VTE and positive findings of PE with COVID-19 infection, we performed additional sensitivity and specificity testing after excluding patients with prior VTE. Within this group and among CTA of the pulmonary arteries only, the sensitivity of the d-dimer was 99.0% using the standard threshold of 500ng/ml, was 98.4% using the age-adjusted d-dimer cutoff, and was 92.1% at an elevated threshold of 1000ng/ml. The NPV of d-dimer was high irrespective of the threshold used: 1000ng/ml (99.2%), age-adjusted cutoff (99.4%) and standard cutoff (99.3%).
Conclusion: This study of COVID-19 positive patients more than doubles the number of patients examining the diagnostic utility of d-dimer in PE diagnosis compared to recent meta-analyses and with a consistent d-dimer methodology performed in close proximity to diagnostic imaging. D-dimer was highly predictive of PE with strong and expected NPVs at standard thresholds and using age-adjusted thresholds. We demonstrate that a higher d-dimer threshold of 1000ng/ml would maintain a very high NPV and overall AUC. A fixed and higher d-dimer threshold in patients with COVID-19 infection with possible PE would significantly reduce the need for CT angiography (by 42%).
Disclosures
Pruthi:Instrumentation Laboratory: Honoraria, Membership on an entity's Board of Directors or advisory committees; HEMA Biologics: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bayer Healthcare AG: Honoraria, Membership on an entity's Board of Directors or advisory committees; Genentech Inc: Honoraria, Membership on an entity's Board of Directors or advisory committees; CSL Behring: Honoraria, Membership on an entity's Board of Directors or advisory committees; Merck: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal